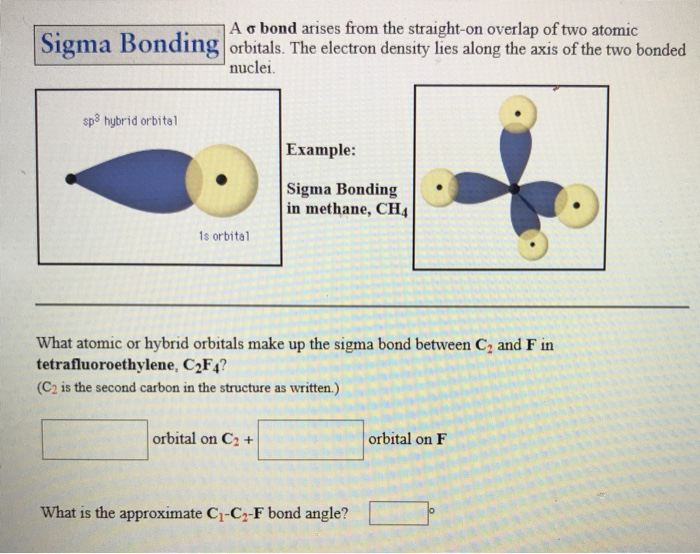 
Sigma bonds are formed by the overlap of orbitals. The type of chemical bond maximizes the stability of the atoms that form it. A sigma bond can be formed by overlap of an s atomic orbital with a p atomic orbital. Sigma bonds are often formed by the combination of s orbitals in. Hybrid orbitals are constructed from valence atomic orbitals and used to make sigma bonds between atoms. Atoms form chemical bonds to make their outer electron shells more stable. This type of bond is referred to as a (sigma) bond. Only head to head overlap is possible with s orbitals because they are spherical. A single bond always contains one sigma bond and a double bond contains one sigma and. Study with Quizlet and memorize flashcards containing terms like Who was the first. POSSIBLE ORBITAL COMBINATIONS TO GENERATE SIGMA MOLECULAR ORBITALS For simplicity, if we examine only the s, p, and d orbitals, let's suppose that all orbitals we are examining are similar enough in energy to interact. Table of contents No headers When atomic orbitals (pure or hybrid) of different atoms overlap to form covalent bonds, they may approach each other in two major ways: head to head, or sideways. Sigma and pi bonds are formed by the overlap of atomic orbitals. This implies that σ-overlap leads to more strongly bonding and anti-bonding orbitals with a larger energy gap between them compared to π-overlap and π-overlap leads to more strongly bonding and anti-bonding orbitals compared to δ-overlap. bonds must be made by orbitals that overlap head-on. This is because in the first case they point toward each other, and the orbital overlap is on the bond axis, while in the latter case they are oriented parallel to each other, and the orbital overlap is above and below the bond axis. Remember that covalent bonds form from the overlap of atomic orbitals which are just the space where electrons are likely to be found.\) Visual representation of orbitals overlapping in σ-fashion vs orbitals overlapping in π-fashion Saturated Unsaturated and Supersaturated The type of hybrid orbitals formed in a bonded atom depends on its electron-pair geometry as predicted by the VSEPR theory.Reaction Quotient and Le Chatelier's Principle.Prediction of Element Properties Based on Periodic Trends.Molecular Structures of Acids and Bases.the 1s orbital of each of the four hydrogen atoms overlaps with one of the four sp 3 orbitals of the carbon atom to form a sigma () bond. 
Orbital overlap can lead to bond formation. The bonds that form by the head-on overlap of orbitals are called (sigma) bonds. End-to-end overlapoverlapIn chemical bonds, an orbital overlap is the concentration of orbitals on adjacent atoms in the same regions of space. Now, there are two types of covalent bonds: sigma () and pi () bonds. A triple bond consists of one pi bond and two sigma bonds. A double bond consists of two pairs of electrons. The p orbital is one orbital that can hold up to two electrons. Bonds formed from atomic s orbitals can be either sigma or Pi bond - Wikipedia. Sigma bonds arent necessarily between two s-orbitals. Ion and Atom Photoelectron Spectroscopy The number of atomic orbitals combined always equals the number of hybrid orbitals formed. functions (orbitals) combine to form hybrid atomic orbitals (sp, sp2, sp3).Elemental Composition of Pure Substances.Application of Le Chatelier's Principle.Structure, Composition
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
